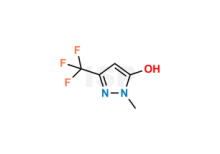
Prednisolone Sodium succinate
| ISP CAT No | |
|---|---|
| CAS No | |
| Mol.F. | |
| Mol.Wt. | |
| Inv. Status | |
| Rel. CAS No |
Disclaimer
The product information provided on this website by ISPStandards is based on the best available understanding at the time of publication. Customers are responsible for verifying the accuracy of this information before making a purchase. ISPStandards may update product details as new information or specifications become available, without prior notice.
- Product Overview
- Description
- Technical Data
Description
Chemical Name: Sodium 4-(2-((8S,9S,10R,11S,13S,14S,17R)-11,17-dihydroxy-10,13-dimethyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-17-yl)-2-oxoethoxy)-4-oxobutanoate
Synonym: Sodium prednisolone 21-hemisuccinate
Shipping Temperature:
Ambient
HSN Code:
38229010
Country of Origin: India
Smiles: O=C1C=C[C@@]2(C)C(CC[C@]3([H])[C@]2([H])[C@](O)([H])C[C@@]4(C)[C@@]3([H])CC[C@]4(O)C(COC(CCC([O-])=O)=O)=O)=C1.[Na+]
Additional Information
Prednisolone Sodium succinate is chemically Sodium 4-(2-((8S,9S,10R,11S,13S,14S,17R)-11,17-dihydroxy-10,13-dimethyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-17-yl)-2-oxoethoxy)-4-oxobutanoate.
It is also known as Sodium prednisolone 21-hemisuccinate .
Prednisolone Sodium succinate is supplied with detailed characterization data compliant with regulatory guideline. Prednisolone Sodium succinate can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Prednisolone.
The product can be used as reference standards and further traceability against pharmacopeial standards (USP or EP) can be provided based on feasibility. ISP Standards products are for analytical purpose only and not for human use.
Download MSDS